A Tasmanian-led study has found that a method of promoting lung expansion and oxygen levels in premature babies decreases the risk of respiratory illness later in life.
Subscribe now for unlimited access.
or signup to continue reading
The international clinical trial, led by University of Tasmania Menzies Institute for Medical Research Professor Peter Dargaville, was conducted in 33 neonatal intensive care units in 11 countries and is the largest study of this type ever conducted.
The trial examined an innovative method for delivering surfactant, a natural product that promotes lung expansion and improves oxygen levels in premature infants.
Professor Dargaville said the study aimed to improve the standard of care on the lives of preterm infants.
The research was on limiting the development of a chronic lung disease bronchopulmonary dysplasia (BPD).
"There was a one-third reduction in the rate of hospitalisation with a respiratory illness, and also reductions in the rate of wheezing or breathing difficulty reported by parents, use of bronchodilator therapy, and a physician report of asthma," he said.
A follow-up study, which reported the two-year outcomes for preterm infants recruited into the trial, was published in the Journal of the American Medical Association (JAMA) on September 11, 2023.
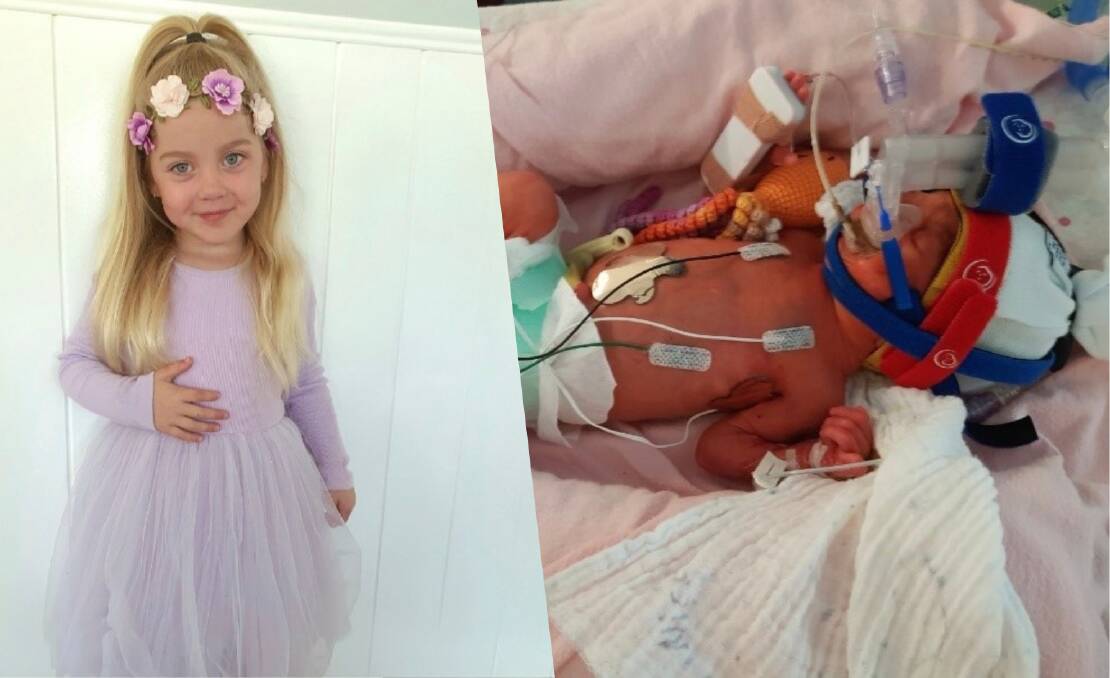
Sarah de Jong's daughter, Eva, was one of the 486 premature babies who took part in the study after her birth at 27 and a half weeks gestation, three months earlier than full term.
Ms de Jong said she didn't hesitate to take part in the trial.
"If this was a chance to help Eva, we gladly accepted," she said.
"We now have an extremely healthy daughter, all thanks to a huge team of people who helped Eva through her journey."
Professor Dargaville said the effectiveness of the trial was demonstrated in the follow-up study.
"We found no differences in neurodevelopmental outcome at two years of age, but a clear difference favouring the active treatment group for respiratory health in the first two years," he said.
"The lasting effect on respiratory health that was found after a single intervention performed in the first hours of life is a major advance and one that cements the place of this new form of surfactant therapy using a thin catheter in preterm infants.
"The fact of there being no difference in neurodevelopmental outcome is also important, as it confirms the safety of the novel method of surfactant delivery."
Why not have your say? Write a letter to the editor here:
Our journalists work hard to provide local, up-to-date news to the community. This is how you can continue to access our trusted content:
- Bookmark www.examiner.com.au
- Make sure you are signed up for our breaking and regular headlines newsletters
- Follow us on Twitter: @examineronline
- Follow us on Instagram: @examineronline
- Follow us on Google News: The Examiner


